Nuclear energy is an ally in reducing greenhouse gas emissions
The atmosphere is essential for life on Earth for many reasons, in particular its oxygen content. It also contains a large number of other gases and chemicals, some as a result of human activity. It is now well established that emissions of certain gases into the atmosphere have a significant and negative impact on the environment.

The greenhouse effect is the overheating of the earth's surface due to an increase in the amount of solar energy that is trapped in the atmosphere, due to certain gases whose presence and proportion have been modified by human activity. The most important greenhouse gases are:
- Carbon dioxide (CO2)
- Methane (CH4)
- Nitrogen oxides (NOx)
- Chlorofluorocarbons (CFC)
- Sulfur hexafluoride (SF6)
The percentages of influence of these gases on the greenhouse effect are in the following order: 55% for CO2, 24% for CFC, 15% for CH4, and 6% for NOX.
In addition to CO2, fossil fuels produce a series of toxic pollutants depending on the type of fuel and the technology used for their combustion: NOX, SO2, organic impurities, and radioactive elements or heavy metals (such as mercury)
Also, the massive use of cars, heating, and excessive electricity consumption is responsible for the emission of CO2 into the atmosphere. Each family can produce up to 5 tonnes of C02 per year.
Certain gases cause ozone depletion at high levels in the atmosphere, limiting the absorption of ultraviolet radiation. This absorption is essential for life on our planet, so the gases that cause this negative effect must be controlled.
Consequences of these emissions
Global warming is unprecedented in the last 10,000 years. Most of it has been observed over the last half of the 20th century and is attributed to the increase in the concentration of greenhouse gases, mainly CO2, from the use of oil, gas, and coal.
A 1,000 MWe coal-fired power plant can release 44,000 tonnes of SO2 and 22,000 tonnes of NOx, into the atmosphere each year, as well as 320,000 tonnes of ash containing up to 400 tonnes of heavy metals.
NOx plays an important role in the destruction of the ozone layer and the occurrence of acid rain as well as contributing to the greenhouse effect. S02 is the cause of acid rain. Substitution of fossil fuel use would reduce its generation by half.
The short-term consequences if urgent action is not taken will be:
- Change in climatic variables: increase in average global temperature, decrease in snow cover, rise in sea level, increase in precipitation, etc.
- Human migratory processes.
- Increase in the number and intensity of storms.
- Forest fires.
- Increased occurrence of floods and flash floods.
- Spread of pests and diseases to new areas due to climate change.
- Loss of agricultural productive capacity.
- Increased erosion.
- Disappearance of numerous species (more than 1 million in the next 50 years).
Nuclear technology, an ally in preserving the ozone layer
The fuels used in the production of non-renewable energy are limited, so we must make rational and efficient use of energy if we do not want to have serious energy supply problems and do without all the technologies we currently use that require electricity to function.
Nuclear power plants are a clean alternative; during their life cycle, they are the technology that emits the least carbon dioxide into the atmosphere in the production of energy.
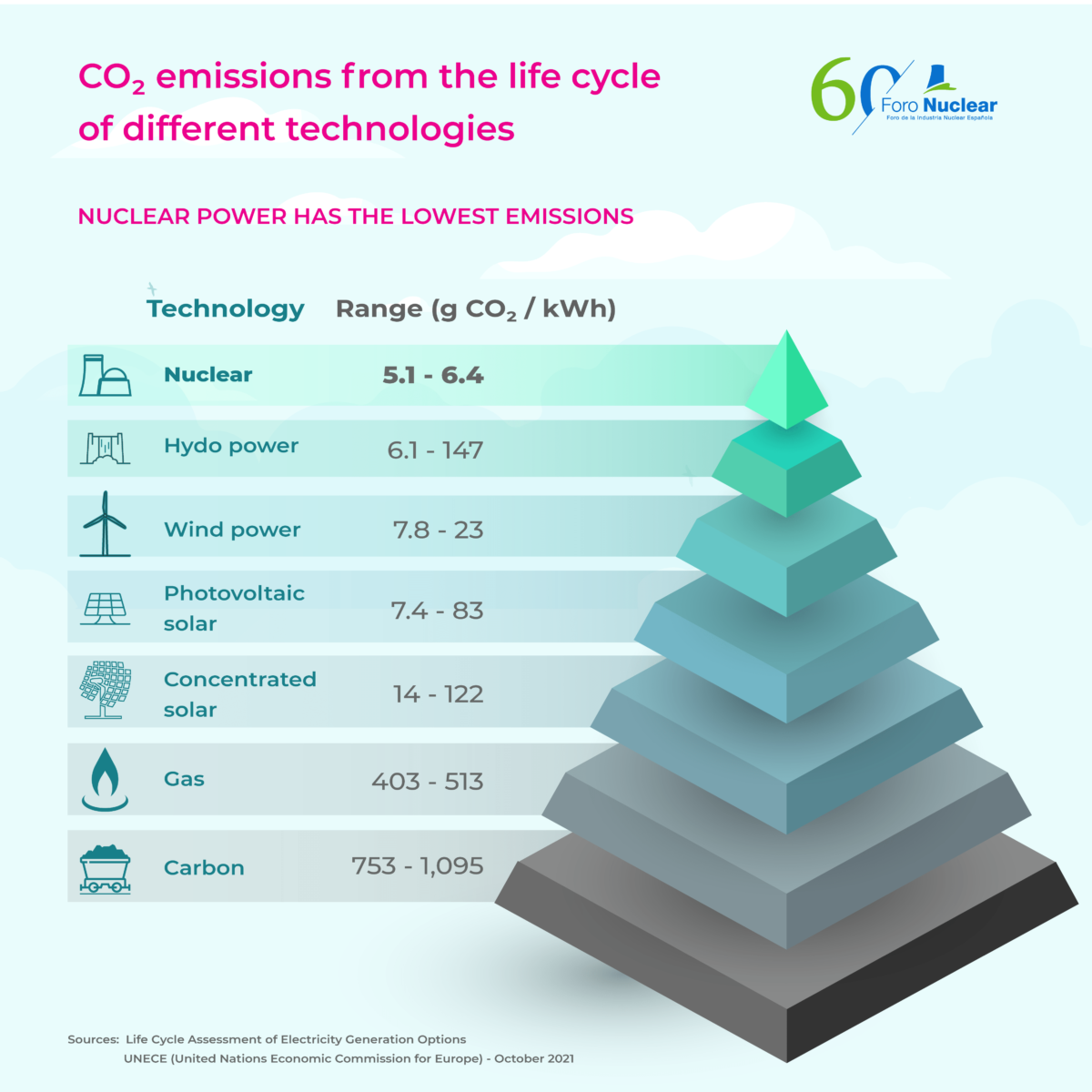
Why? Because a nuclear power plant produces electrical energy through a physical process, the fission of the uranium atom, and not through combustion. Therefore, in its operation, no greenhouse gases or other combustion products are emitted into the atmosphere, and only very low carbon dioxide emissions when considering its entire life cycle (from construction to decommissioning).
For this reason, nuclear energy is an important technological option in climate change mitigation strategies and in the preservation of the ozone layer.


