Wolfgang Ernst Pauli promulgated the Exclusion Principle, introduced a fourth quantum number, and theorized about the neutrino

April 25, 1900, in Zurich (Switzerland) – December 15, 1958, in Zurich (Switzerland)
Wolfgang Ernest Paulí graduated in Physics from the Döblinger Gymnasium in Vienna (Austria) in 1918 and, just two months later, he published his first article on Albert Einstein's theory of general relativity. In 1921, he obtained a Ph.D. in Physics from the University of Munich (Germany), tutored by Arnold Sommerfeld, a German physicist specializing in studies of the atom.
At Einstein's suggestion, he published a 237-page monograph on relativity in the German journal Encyklopaedie der mathematischen Wissenschaften ("Encyclopedia of Mathematical Sciences"), receiving praise from Einstein and which remains one of the basic references on the subject today. .
He spent a year at the University of Göttingen (Germany) as an assistant to Max Born, learning all the secrets of Quantum Mechanics, a branch of contemporary Physics in which Born was a world authority. A year later, he moved to the Institute of Theoretical Physics in Copenhagen (Denmark) to collaborate with Niels Bohr, another physicist who deeply marked Pauli's career.
Between 1923 and 1928, he served as Professor of Theoretical Physics at the University of Hamburg. It is during this time that he developed his famous Exclusion Principle and his other non-relativistic theory of Spin.
In 1928, he left Germany to occupy the Chair of Theoretical Physics at the Federal Institute of Technology (ETH) in Zurich (Switzerland).
In 1931 and 1935, he moved to the United States to teach seminars at the University of Michigan and the Institute for Advanced Study at Princeton, respectively.
In this period of time, in 1932, Pauli made his third great contribution to science, the theoretical proposal of the existence of the neutrino, a subatomic particle that could offer a satisfactory explanation for the beta decay of radium (although it was not experimentally detected until 1956). ).
In 1933, he published "The Fundamental Principles of Quantum Mechanics", considered one of the fundamental texts of this subject.
In 1938, Hitler annexed Austria so he forcibly became a German citizen. After the outbreak of World War II he moved to the United States to take over the chair of Physics at Princeton.
In 1945, he received the Nobel Prize in Physics for his discovery of the Exclusion Principle and, a year later, he received American citizenship.
After the war, he returned to Zurich until his death at the age of 58.
The spin
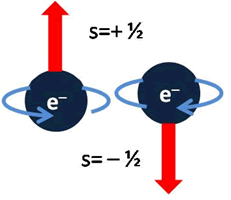
In the beginning, quantum theory recognized the existence of three quantum numbers: the main one (which, according to Niels Bohr, defines the energy of the electron in its orbit), the magnetic one (which marks the orientation of the orbit in space) and the azimuthal (describing the shape of the elliptical orbit).
Traditional Physics accepted the existence of these numbers, but was suspicious of Quantum Mechanics because, with them, it was impossible to explain the spectrum of the atom.
To solve this problem, in 1924, Pauli added a fourth number, the spin, which represented the measure of the angular momentum of the electron or its direction of spin on itself and established the only two values that it can take: ½ and – ½.
Pauli exclusion principle
In 1925, Pauli enunciated this principle by virtue of which there cannot be, in the same atom, two electrons with the same value for the four quantum numbers. Therefore, electrons, electrically charged particles that move around the atomic nucleus, are mutually exclusive (since they cannot overlap each other in the same orbit because they repel each other).
The space within an atom in which the electrons are found and cannot remain at the same time if they are not whole and have the same arrangement is called an orbital.
Thanks to this principle, it was possible to explain the arrangement of the elements in the periodic table, as well as to establish the orbits of their electrons. Later it was discovered that it could also be applied to all elementary particles encompassed within the group of fermions (those whose spin is not an integer).
The neutrino

A phenomenon difficult to explain had been detected, the so-called beta decay. It was a discrepancy of moments and energies that resulted in the emission or ejection of electrons by the atomic nucleus. The strange thing is that, in its very nature, it contradicted the principle of conservation of energy.
Pauli made a theoretical proposal about this phenomenon in which he indicated the existence of an extremely light neutral particle, not detected experimentally until then, whose emission coincided with that of the electron. Said particle arose at the moment of beta disintegration, taking the energy and the moment that were missing; but since it had zero or practically zero mass, it barely interacted with matter, making it very difficult to detect.
The Italian physicist Enrico Fermi supported this theory by baptizing the particle with the name of neutrino and, in 1956, Frederick Reines managed to detect it experimentally, giving full validity to Pauli's theoretical speculation. Subsequently, scientists such as Lederman, Schwaartz and Steinberger revealed the existence of various types of neutrinos.
What is the Pauli effect?
In Spain, known as the “demo effect”, it refers to the apparently mysterious breakdowns of technical equipment in the presence of certain people. This happened to Pauli, that's why he was named after him. However, Wolfgang Ernest Pauli, aware of his reputation, was not offended and found it rather amusing whenever this effect (not to be confused with the type of physical phenomenon that is his Exclusion Principle) manifested itself.

