Sommerfeld's atomic model (1916 AD)
Temas: Periodic Tables | Science

It was proposed by Arnold Sommerfield to try to cover the deficiencies that the Bohr model presented. Therefore, it is a relativistic version of the Rutherford-Bohr model and was based, in part, on Albert Einstein's relativistic postulates.
Bohr's atomic model was seamless when it came to the hydrogen atom. However, when it came to atoms of other chemical elements, the electrons of the same energy level had different energy.
Therefore, he asserted a number of corrections to the model:
He discovered that the electrons of certain atoms reached speeds close to that of light. He performed the calculations for relativistic electrons.
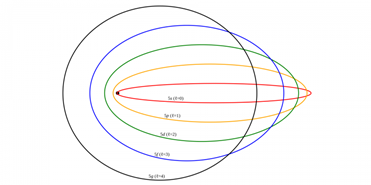
- Electrons move around the nucleus of the atom, in circular or elliptical orbits.
- From the second energy level there is one or more sublevels on the same level with slightly different energies.
- The eccentricity of the orbit gave rise to a new quantum number that determines the shape of orbitals: the azimuthal quantum number.
- Electrons have tiny electrical currents.
Tipos: Theory
Edades: +16 years old | 06-12 years old | 12-16 years old | All ages
Etiquetas: Atoms - periodic table

