Schrödinger's atomic model (1926 AD)

It is known as the "Quantum-Wave Model" and was proposed by Erwin Schrödinger, in 1926, based on the studies of De Broglie, Bohr and Sommerfeld.
His model conceives of electrons as undulations of matter, that is, it describes the wave behavior of the electron.
Schrödinger suggested that the movement of electrons in the atom corresponded to wave-particle duality, and consequently electrons could move around the nucleus as standing waves.
This allowed the later formulation of a probabilistic interpretation of the wave function (magnitude used to describe the probability of finding a particle in space) by Max Born and meant that the position of an electron or its path could be studied probabilistically. momentum but not both, due to the Heisenberg Uncertainty Principle.
Schrödinger was awarded the Nobel Prize, in 1933, for his contributions to atomic theory, the development of the equation to calculate the probability that an electron is in a specific position.
Therefore, the characteristics of your model are:
- Describe the movement of electrons as standing waves.
- Electrons are constantly moving, that is, they do not have a fixed or defined position within the atom.
- It does not predict the location of the electron, nor does it describe the path it takes within the atom. It only establishes a probability zone to locate the electron.
- These probability areas are called atomic orbitals. Orbitals describe a translation movement around the nucleus of the atom.
- These atomic orbitals have different energy levels and sub-levels, and can be defined between electron clouds.
- The model does not contemplate the stability of the nucleus, it only refers to explaining the quantum mechanics associated with the movement of electrons within the atom.

In the image you can see the electron density that indicates the probability of finding an electron near the nucleus. The closer it is to the nucleus (purple zone) the more probability there will be, while it will be less if it is further away from the nucleus (purple zone).
Schrodinger's equation
Schrödinger developed two mathematical models, differentiating what happens depending on whether the quantum state changes with time or not and, at the end of 1926, published the time-independent equation that bears his name, which is based on the fact that wave functions behave like standing waves.
This implies that the wave does not move, its nodes, that is, its equilibrium points, serve as a pivot for the rest of the structure to move around them, describing a determined frequency and amplitude.
Schrödinger defined the waves described by electrons as stationary or orbital states, and they are associated, in turn, with different energy levels.
The time-independent Schrödinger equation is as follows:
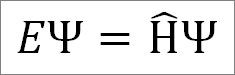
Where:
E: constant of proportionality.
Ψ: wave function of the quantum system.
Η ̂: Hamiltonian operator.
Applied to the Schrödinger atomic model, if the electron moves in a defined space, there are discrete energy values, and if the electron moves freely in space, there are continuous energy intervals.
From the mathematical point of view, there are several solutions for the Schrödinger equation, each solution implies a different value for the constant of proportionality E.
Applying the postulates of the Schrödinger atomic model to this equation, we have:
- Electrons behave like standing waves that are distributed in space according to the wave function Ψ.
- Electrons move within the atom in describing orbitals. These are areas where the probability of finding an electron is considerably higher. The referred probability is proportional to the square of the wave function Ψ2.
- The electronic configuration of the Schrödinguer atomic model explains the periodic properties of atoms and the bonds they form.
However, Schrödinger's atomic model does not take into account the spin of electrons, nor does it take into account variations in the behavior of fast electrons due to relativistic effects.


