Radiation Basics

What is radiation?
Radiation is nothing more than the emission, propagation and transfer of energy in any medium in the form of electromagnetic waves or particles.
How is electromagnetic radiation characterized?
The energy carried by electromagnetic radiation travels through waves. This energy is not continuous, but is transmitted grouped into small "quanta" of energy called photons. The wave is composed of an oscillating electric field, associated with an oscillating magnetic field, and both are perpendicular to the direction of propagation.
Electromagnetic waves are characterized by three fundamental parameters: Wavelength, Frequency and Energy:
- Wavelength (λ): It is the distance between the crests of two consecutive waves, and is measured in units of length (m). The amplitude of the wave depends on the radiant power of the emitting source.
- Frequency (ν or f): It is the number of times a wave oscillates in one second and is measured in cycles/second or hertz (Hz).
- Energy (E): The energy carried by electromagnetic radiation can be measured in Joules (J), although it is more frequently measured in electronvolts (eV). The electronvolt represents the kinetic energy that an electron acquires when it is accelerated by a potential difference of 1 volt. It is equivalent to 1.602176462 × 10-19 J.
The frequency is directly proportional to the energy that radiation carries, according to the equation: E = h.f, (where h is Planck's constant = 6.63·10-34 J/s).
The different electromagnetic radiations have different energies and interact with matter in a different way depending on this energy.
The following diagram shows the different types of electromagnetic radiation ordered by energy. It is what is known as the electromagnetic spectrum. Surely many of these radiations are familiar to you.
Electromagnetic radiations are classified into two large groups based on their energy, or in other words based on the type of changes they cause in the atoms with which they interact:
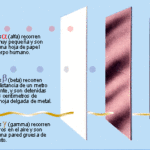
- Ionizing radiation. They correspond to the highest energy radiations (shortest wavelength) within the electromagnetic spectrum. They have enough energy to remove electrons from the atoms with which they interact, that is, to produce ionizations.
- Non-ionizing radiation. These radiations do not have enough energy to produce ionizations in the atoms with which they interact, hence their name.
Of all the electromagnetic radiation shown in the previous diagram (electromagnetic spectrum), only X-rays and gamma rays have enough energy to produce ionization phenomena in atoms, that is, they are ionizing radiation. The rest of the electromagnetic radiations (radio waves, microwaves, infrared rays, visible light, ultraviolet rays) are non-ionizing radiations.