Dirac-Jordan model of the atom (1928 AD)

Also called “quantum-mechanical” model, unlike the Schrödinger model, it is not necessary to impose the spin by means of the Pauli exclusion principle, since it appears naturally.
In addition, it incorporates the relativistic corrections of the spin-orbit interaction and the Darwin term, which account for the fine structure of the electronic levels of the atom.
In 1928, the scientists Paul A. M. Dirac and Pascual Jordan set out to generalize the quantum mechanics developed by Schrödinger to include Einstein's special relativity corrections.
Dirac starts from Schrodinger's equation, which consists of a differential operator, called a Hamiltonian, that operates on a function known as the electron wave function. However Schrodinger did not take relativistic effects into account.
The solutions of the wave function allow us to calculate the regions where with a certain degree of probability the electron will be found around the nucleus. These regions or zones are called orbitals and depend on certain discrete quantum numbers, which define the energy and angular momentum of the electron.
In the image you can see the electronic orbitals of the hydrogen atom for the first three energy levels:
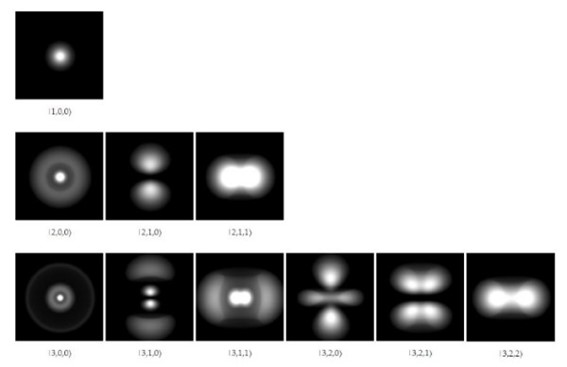
In quantum mechanical theories, whether relativistic or not, there is no concept of orbits, since neither the position nor the speed of the electron can be specified simultaneously. And furthermore, specifying one of the variables leads to a total imprecision in the other.
In the Dirac-Jordan equations, the fourth parameter with quantum characteristic appears, called s, in addition to the already known n, l and m.

